Best of: FDA moves to make it easier for men who have sex with men to give blood
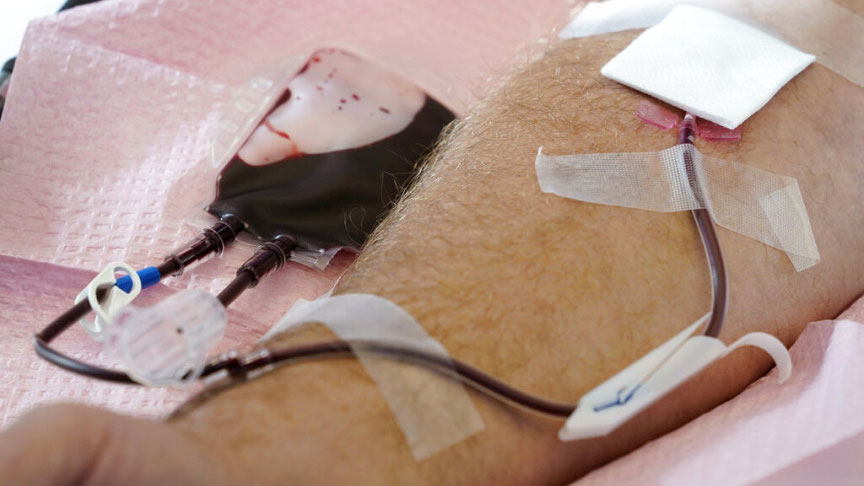
Tubes direct blood from a donor into a bag in Davenport, Iowa, on Friday, Nov. 11, 2022. The U.S. is moving to ease restrictions on blood donations from gay and bisexual men and other groups that traditionally face higher risks of HIV. TThe Food and Drug Administration on Friday, Jan. 27, 2023, announced draft guidelines that would do away with the current three-month abstinence requirement for donations from men who have sex with men. Charlie Neibergall/AP
Since it first issued a lifetime ban on blood donations by men who have sex with men (MSM) in 1985 over concerns about HIV transmission, the FDA has changed its policy on this issue twice. First, in 2015, it lifted the lifetime ban and imposed a one-year deferral period, requiring MSM to be abstinent for 12 months before they could donate. Then, in April of 2020, the administration shortened the waiting period to three months. Now, it has eliminated that time restriction altogether and shifted to individual HIV risk assessments for all donors.
To talk about what's in the FDA's newly updated guidelines, the science behind them, and the stigma they have caused, we were joined by a doctor who specializes in infectious diseases, an LGBTQ advocate, and a representative from a Midwestern blood bank.
This conversation originally aired Feb. 9, 2023.
GUESTS:
Dr. Katharine Bar
Assistant Professor of Medicine, Perelman School of Medicine | Attending physician in infectious diseases, University of Pennsylvania Hospital | Co-author, “Science over stigma: the need for evidence-based blood donation policies for men who have sex with men in the USA”
Jerry Carden
Founding Member, Greater Community AIDS Project of Champaign
Amanda Hess
Vice President for Donor Relations and Marketing, ImpactLife
Prepared for web by Owen Henderson
Help shape our coverage on The 21st by joining our texting group and answering weekly questions. To join, text “TALK” to 217-803-0730 or sign up with your phone number below:

